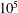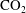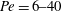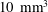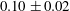12 results
A continuum-scale representation of Ostwald ripening in heterogeneous porous media
- Yaxin Li, Charlotte Garing, Sally M. Benson
-
- Journal:
- Journal of Fluid Mechanics / Volume 889 / 25 April 2020
- Published online by Cambridge University Press:
- 21 February 2020, A14
-
- Article
- Export citation
-
Ostwald ripening is a pore-scale phenomenon that coarsens a dispersed phase until thermodynamic equilibrium. Based on our previous finding that multi-bubble equilibrium is possible and likely in complex porous media, we develop a new continuum-scale model for Ostwald ripening in heterogeneous porous media. In this model, porous media with two different capillary pressure curves are put into contact, allowing only diffusive flow through the aqueous phase to redistribute a trapped gas phase. Results show that Ostwald ripening can increase the gas saturation in one medium while decreasing the gas saturation in the other, even when the gas phase is trapped in pore spaces by capillary forces. We develop an analogous retardation factor to show that the characteristic time for Ostwald ripening is about
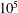 $10^{5}$ times slower than a single-phase diffusion problem due to the fact that separate-phase gas requires a much larger amount of mass transfer before equilibrium is established. An approximate solution has been developed to predict the saturation redistribution between the two media. The model has been validated by numerical simulation over a wide range of physical parameters. Millimetre to centimetre-scale systems come to equilibrium in years, ranging up to 10 000 years and longer for metre-scale systems. These findings are particularly relevant for geological
$10^{5}$ times slower than a single-phase diffusion problem due to the fact that separate-phase gas requires a much larger amount of mass transfer before equilibrium is established. An approximate solution has been developed to predict the saturation redistribution between the two media. The model has been validated by numerical simulation over a wide range of physical parameters. Millimetre to centimetre-scale systems come to equilibrium in years, ranging up to 10 000 years and longer for metre-scale systems. These findings are particularly relevant for geological  $\text{CO}_{2}$ storage, where residual trapping is an important mechanism for immobilizing
$\text{CO}_{2}$ storage, where residual trapping is an important mechanism for immobilizing 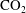 $\text{CO}_{2}$. Our work demonstrates that Ostwald ripening due to heterogeneity in porous media is slow and on a similar time scale compared to other processes that redistribute trapped
$\text{CO}_{2}$. Our work demonstrates that Ostwald ripening due to heterogeneity in porous media is slow and on a similar time scale compared to other processes that redistribute trapped  $\text{CO}_{2}$ such as convective mixing.
$\text{CO}_{2}$ such as convective mixing.
Pore-scale modelling of Ostwald ripening – CORRIGENDUM
- Jacques A. de Chalendar, Charlotte Garing, Sally M. Benson
-
- Journal:
- Journal of Fluid Mechanics / Volume 866 / 10 May 2019
- Published online by Cambridge University Press:
- 18 March 2019, p. 929
-
- Article
-
- You have access Access
- HTML
- Export citation
Pore-scale modelling of Ostwald ripening
- Jacques A. de Chalendar, Charlotte Garing, Sally M. Benson
-
- Journal:
- Journal of Fluid Mechanics / Volume 835 / 25 January 2018
- Published online by Cambridge University Press:
- 27 November 2017, pp. 363-392
-
- Article
-
- You have access Access
- HTML
- Export citation
-
In a saturated solution with dispersed clusters of a second phase, the mechanism by which the larger clusters grow at the expense of the smaller ones is called Ostwald ripening. Although the mechanism is well understood in situations where multiple clusters of gas exist in a liquid solution, evolution is much more complicated to predict when the two phases interact with a solid matrix, since the solid plays a significant role in determining the shape of the interface. In this paper, we study capillary dominated regimes in porous media where the driving force is inter-cluster diffusion. By decomposing the Ostwald ripening mechanism into two processes that operate on different time scales – the diffusion of solute gas in the liquid and the readjustment of the shape of the gas–liquid interface to accommodate a change in mass – we develop a sequential algorithm to solve for the evolution of systems with multiple gas ganglia. In the absence of a solid matrix, thermodynamic equilibrium is reached when all of the gas phase aggregates to form one large bubble. In porous media on the other hand, we find that ripening can lead to equilibrium situations with multiple disconnected ganglia, and that evolution is highly dependent on initial conditions and the structure of the solid matrix. The fundamental difference between the two cases is in the relationship between mass and capillary pressure.
Quantifying solute spreading and mixing in reservoir rocks using 3-D PET imaging
- Ronny Pini, Nicholas T. Vandehey, Jennifer Druhan, James P. O’Neil, Sally M. Benson
-
- Journal:
- Journal of Fluid Mechanics / Volume 796 / 10 June 2016
- Published online by Cambridge University Press:
- 10 May 2016, pp. 558-587
-
- Article
- Export citation
-
We report results of an experimental investigation into the effects of small-scale (mm–cm) heterogeneities on solute spreading and mixing in a Berea sandstone core. Pulse-tracer tests have been carried out in the Péclet number regime
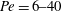 $Pe=6{-}40$ and are supplemented by a unique combination of two imaging techniques. X-ray computed tomography (CT) is used to quantify subcore-scale heterogeneities in terms of permeability contrasts at a spatial resolution of approximately
$Pe=6{-}40$ and are supplemented by a unique combination of two imaging techniques. X-ray computed tomography (CT) is used to quantify subcore-scale heterogeneities in terms of permeability contrasts at a spatial resolution of approximately 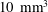 $10~\text{mm}^{3}$, while [11C] positron emission tomography (PET) is applied to image the spatial and temporal evolution of the full tracer plume non-invasively. To account for both advective spreading and local (Fickian) mixing as driving mechanisms for solute transport, a streamtube model is applied that is based on the one-dimensional advection–dispersion equation. We refer to our modelling approach as semideterministic, because the spatial arrangement of the streamtubes and the corresponding solute travel times are known from the measured rock’s permeability map, which required only small adjustments to match the measured tracer breakthrough curve. The model reproduces the three-dimensional PET measurements accurately by capturing the larger-scale tracer plume deformation as well as subcore-scale mixing, while confirming negligible transverse dispersion over the scale of the experiment. We suggest that the obtained longitudinal dispersivity (
$10~\text{mm}^{3}$, while [11C] positron emission tomography (PET) is applied to image the spatial and temporal evolution of the full tracer plume non-invasively. To account for both advective spreading and local (Fickian) mixing as driving mechanisms for solute transport, a streamtube model is applied that is based on the one-dimensional advection–dispersion equation. We refer to our modelling approach as semideterministic, because the spatial arrangement of the streamtubes and the corresponding solute travel times are known from the measured rock’s permeability map, which required only small adjustments to match the measured tracer breakthrough curve. The model reproduces the three-dimensional PET measurements accurately by capturing the larger-scale tracer plume deformation as well as subcore-scale mixing, while confirming negligible transverse dispersion over the scale of the experiment. We suggest that the obtained longitudinal dispersivity (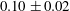 $0.10\pm 0.02$ cm) is rock rather than sample specific, because of the ability of the model to decouple subcore-scale permeability heterogeneity effects from those of local dispersion. As such, the approach presented here proves to be very valuable, if not necessary, in the context of reservoir core analyses, because rock samples can rarely be regarded as ‘uniformly heterogeneous’.
$0.10\pm 0.02$ cm) is rock rather than sample specific, because of the ability of the model to decouple subcore-scale permeability heterogeneity effects from those of local dispersion. As such, the approach presented here proves to be very valuable, if not necessary, in the context of reservoir core analyses, because rock samples can rarely be regarded as ‘uniformly heterogeneous’.
Summary for Policy Makers
-
- By Thomas B. Johansson, Lund University, Nebojsa Nakicenovic, International Institute for Applied Systems Analysis and Vienna University of Technology, Anand Patwardhan, Indian Institute of Technology-Bombay), Luis Gomez-Echeverri, International Institute for Applied Systems Analysis, Rangan Banerjee, Indian Institute of Technology, Sally M. Benson, Stanford University, Daniel H. Bouille, Bariloche Foundation, Abeeku Brew-Hammond, Kwame Nkrumah University of Science and Technology, Aleh Cherp, Central European University, Suani T. Coelho, National Reference Center on Biomass, University of São Paulo, Lisa Emberson, Stockholm Environment Institute, University of York, Maria Josefina Figueroa, Technical University, Arnulf Grubler, International Institute for Applied Systems Analysis, Austria and Yale University, Kebin He, Tsinghua University, Mark Jaccard, Simon Fraser University, Suzana Kahn Ribeiro, Federal University of Rio de Janeiro, Stephen Karekezi, AFREPREN/FWD, Eric D. Larson, Princeton University and Climate Central, Zheng Li, Tsinghua University, Susan McDade, United Nations Development Programme), Lynn K. Mytelka, United Nations University-MERIT, Shonali Pachauri, International Institute for Applied Systems Analysis, Keywan Riahi, International Institute for Applied Systems Analysis, Johan Rockström, Stockholm Environment Institute, Stockholm University, Hans-Holger Rogner, International Atomic Energy Agency, Joyashree Roy, Jadavpur University, Robert N. Schock, World Energy Council, UK and Center for Global Security Research, Ralph Sims, Massey University, Kirk R. Smith, University of California, Wim C. Turkenburg, Utrecht University, Diana Ürge-Vorsatz, Central European University, Frank von Hippel, Princeton University, Kurt Yeager, Electric Power Research Institute and Galvin Electricity Initiative
- Global Energy Assessment Writing Team
-
- Book:
- Global Energy Assessment
- Published online:
- 05 September 2012
- Print publication:
- 27 August 2012, pp 3-30
-
- Chapter
- Export citation
-
Summary
Introduction
Energy is essential for human development and energy systems are a crucial entry point for addressing the most pressing global challenges of the 21st century, including sustainable economic and social development, poverty eradication, adequate food production and food security, health for all, climate protection, conservation of ecosystems, peace and security. Yet, more than a decade into the 21st century, current energy systems do not meet these challenges.
A major transformation is therefore required to address these challenges and to avoid potentially catastrophic future consequences for human and planetary systems. The Global Energy Assessment (GEA) demonstrates that energy system change is the key for addressing and resolving these challenges. The GEA identifies strategies that could help resolve the multiple challenges simultaneously and bring multiple benefits. Their successful implementation requires determined, sustained and immediate action.
Transformative change in the energy system may not be internally generated; due to institutional inertia, incumbency and lack of capacity and agility of existing organizations to respond effectively to changing conditions. In such situations clear and consistent external policy signals may be required to initiate and sustain the transformative change needed to meet the sustainability challenges of the 21st century.
The industrial revolution catapulted humanity onto an explosive development path, whereby, reliance on muscle power and traditional biomass was replaced mostly by fossil fuels. In 2005, some 78% of global energy was based on fossil energy sources that provided abundant and ever cheaper energy services to more than half the people in the world.
Chapter 13 - Carbon Capture and Storage
-
- By Sally M. Benson, Stanford University, Kamel Bennaceur, Schlumberger, Peter Cook, Cooperative Research Centre for Greenhouse Gas Technologies, John Davison, IEA Greenhouse Gas R&D Programme, Heleen de Coninck, Energy research Centre of the Netherlands, Karim Farhat, Stanford University, Andrea Ramirez, Utrecht University, Dale Simbeck, SFA Pacific, Terry Surles, Desert Research Institute, Preeti Verma, The Climate Group, Iain Wright, John Ahearne, Sigma Xi
- Global Energy Assessment Writing Team
-
- Book:
- Global Energy Assessment
- Published online:
- 05 September 2012
- Print publication:
- 27 August 2012, pp 993-1068
-
- Chapter
- Export citation
-
Summary
Executive Summary
Emissions of carbon dioxide, the most important long-lived anthropogenic greenhouse gas, can be reduced by Carbon Capture and Storage (CCS). CCS involves the integration of four elements: CO2 capture, compression of the CO2 from a gas to a liquid or a denser gas, transportation of pressurized CO2 from the point of capture to the storage location, and isolation from the atmosphere by storage in deep underground rock formations. Considering full life-cycle emissions, CCS technology can reduce 65–85% of CO2 emissions from fossil fuel combustion from stationary sources, although greater reductions may be possible if low emission technologies are applied to activities beyond the plant boundary, such as fuel transportation.
CCS is applicable to many stationary CO2 sources, including the power generation, refining, building materials, and the industrial sector. The recent emphasis on the use of CCS primarily to reduce emissions from coal-fired electricity production is too narrow a vision for CCS.
Interest in CCS is growing rapidly around the world. Over the past decade there has been a remarkable increase in interest and investment in CCS. Whereas a decade ago, there was only one operating CCS project and little industry or government investment in R&D, and no financial incentives to promote CCS. In 2010, numerous projects of various sizes are active, including at least five large-scale full CCS projects. In 2015, it is expected that 15 large-scale, full-chain CCS projects will be running. Governments and industry have committed over USD 26 billion for R&D, scale-up and deployment.
Technical Summary
-
- By Thomas B. Johansson, Lund University, Nebojsa Nakicenovic, International Institute for Applied Systems Analysis and Vienna University of Technology, Anand Patwardhan, Indian Institute of Technology, Luis Gomez-Echeverri, International Institute for Applied Systems Analysis, Doug J. Arent, National Renewable Energy Laboratory, Rangan Banerjee, Indian Institute of Technology, Sally M. Benson, Stanford University, Daniel H. Bouille, Bariloche Foundation, Abeeku Brew-Hammond, Kwame Nkrumah University of Science and Technology, Aleh Cherp, Central European University, Suani T. Coelho, National Reference Center on Biomass, University of São Paulo, Lisa Emberson, Stockholm Environment Institute, University of York, Maria Josefina Figueroa, Technical University, Arnulf Grubler, International Institute for Applied Systems Analysis, Austria and Yale University, Kebin He, Tsinghua University, Mark Jaccard, Simon Fraser University, Suzana Kahn Ribeiro, Federal University of Rio de Janeiro, Stephen Karekezi, AFREPREN/FWD, Eric D. Larson, Princeton University and Climate Central, Zheng Li, Tsinghua University, Susan McDade, United Nations Development Programme, Lynn K. Mytelka, United Nations University-MERIT, Shonali Pachauri, International Institute for Applied Systems Analysis, Keywan Riahi, International Institute for Applied Systems Analysis, Johan Rockström, Stockholm Environment Institute, Stockholm University, Hans-Holger Rogner, International Atomic Energy Agency, Joyashree Roy, Jadavpur University, Robert N. Schock, World Energy Council, UK and Center for Global Security Research, Ralph Sims, Massey University, Kirk R. Smith, University of California, Wim C. Turkenburg, Utrecht University, Diana Ürge-Vorsatz, Central European University, Frank von Hippel, Princeton University, Kurt Yeager, Electric Power Research Institute and Galvin Electricity Initiative
- Global Energy Assessment Writing Team
-
- Book:
- Global Energy Assessment
- Published online:
- 05 September 2012
- Print publication:
- 27 August 2012, pp 31-94
-
- Chapter
- Export citation
-
Summary
Introduction
Energy is essential for human development and energy systems are a crucial entry point for addressing the most pressing global challenges of the 21st century, including sustainable economic, and social development, poverty eradication, adequate food production and food security, health for all, climate protection, conservation of ecosystems, peace, and security. Yet, more than a decade into the 21st century, current energy systems do not meet these challenges.
In this context, two considerations are important. The first is the capacity and agility of the players within the energy system to seize opportunities in response to these challenges. The second is the response capacity of the energy system itself, as the investments are long-term and tend to follow standard financial patterns, mainly avoiding risks and price instabilities. This traditional approach does not embrace the transformation needed to respond properly to the economic, environmental, and social sustainability challenges of the 21st century.
A major transformation is required to address these challenges and to avoid potentially catastrophic consequences for human and planetary systems. The GEA identifies strategies that could help resolve the multiple challenges simultaneously and bring multiple benefits. Their successful implementation requires determined, sustained, and immediate action.
The industrial revolution catapulted humanity onto an explosive development path, whereby reliance on muscle power and traditional biomass was replaced mostly by fossil fuels. In 2005, approximately 78% of global energy was based on fossil energy sources that provided abundant and ever cheaper energy services to more than half the world's population.
3 - Sustainability and energy conversions
- from Part 1 - Energy and the environment: the global landscape
-
- By Franklin M. Orr, Sally M. Benson, Department of Energy Resources Engineering, Stanford University, Stanford, CA, USA
- Edited by David S. Ginley, National Renewable Energy Laboratory, Colorado, David Cahen, Weizmann Institute of Science, Israel
-
- Book:
- Fundamentals of Materials for Energy and Environmental Sustainability
- Published online:
- 05 June 2012
- Print publication:
- 30 November 2011, pp 36-47
-
- Chapter
- Export citation
-
Summary
Focus
Energy use is inexorably woven into the fabric of modern civilization. Human well-being, economic productivity, and national security all depend on the availability of plentiful and affordable energy supplies. However, over the past half century, we have come to understand that continued growth of energy use along the lines of current energy systems will lead to unacceptable consequences for the Earth's climate and oceans. Maintaining and increasing the access to energy services to satisfy crucial societal needs requires the development of a sustainable global energy system that transitions away from energy supply options with high greenhouse gas (GHG) emissions and unhealthy air pollutants. Disparity in energy access is also not sustainable. We must provide sufficient energy for the estimated 1.6 billion people who do not have access to modern energy systems today. Fortunately, plentiful energy resources are available to meet our needs, and technology pathways for making this transition exist. Continuing to lower the cost and increase the reliability of energy from sustainable energy resources will facilitate this transition. Changing the world's energy systems to reduce GHG emissions is one of the critical challenges that humans must face in this century. The required transition can begin now with improvements in efficiency of energy conversion and use, and with continuing deployment over the coming decades of a variety of existing and innovative technologies. With continuing attention to energy conversions that minimize wastes, have low life-cycle impacts, and maximize recycling of materials, a set of sustainable energy systems can be created.
Synopsis
Feeding, clothing, and housing a growing world population will be a significant challenge in this century, as will supplying the fresh water, heat, lighting, and transportation we will need to live comfortable and productive lives. This chapter discusses energy sustainability, with emphasis on the requirement to reduce GHG emissions. A sustainable energy system is one in which energy is supplied and converted to energy services in ways that avoid unacceptable consequences for local, regional, and global natural systems that control climate and support ecosystems that provide essential services. Figure 3.1 illustrates typical conversions of a primary energy resource (solar, wind, geothermal energy, fossil or nuclear resources, etc.) into a product, such as a fuel or an energy carrier, like electricity, that then can be converted to a service like heat, light, or mechanical work. Sustainable processes and systems that convert some primary energy resource into energy services will be ones that are as efficient as possible – smaller quantities of the primary energy resource are needed and fewer waste materials are created if the conversions are efficient. Some have argued that only energy flows such as solar, wind, and wave power should be considered sustainable. Others note that any system of energy conversions has some footprint and impact, and that sustainability is necessarily a relative measure, not an absolute one. In any case, sustainable systems will have low impacts over the full life cycle of the conversions (see Chapter 41). Recycling of materials used in energy conversions will be maximized, and amounts of waste materials created in the chain of energy conversions to services will be minimized when the whole cycle from primary resource to services (such as mechanical work) to waste heat and products is considered.
8 - Carbon dioxide capture and sequestration
- from Part 1 - Energy and the environment: the global landscape
-
- By Sally M. Benson, Department of Energy Resources Engineering, Stanford University, Stanford, CA, USA
- Edited by David S. Ginley, National Renewable Energy Laboratory, Colorado, David Cahen, Weizmann Institute of Science, Israel
-
- Book:
- Fundamentals of Materials for Energy and Environmental Sustainability
- Published online:
- 05 June 2012
- Print publication:
- 30 November 2011, pp 90-104
-
- Chapter
- Export citation
-
Summary
Focus
A transition to a low-carbon economy can be facilitated by CO2 capture and sequestration. This chapter focuses on capture of carbon dioxide from industrial emission sources such as electricity generation and sequestration in deep geological formations. A detailed description of the technology is provided, including the potential scale of application, estimated costs, assessment of risks, and emerging research issues.
Synopsis
Today, 60% of global CO2 emissions come from large point sources such as power plants, refineries, cement plants, and steel mills. Reducing emissions from these sources will require reducing demand for the services or materials they provide, finding alternative ways to provide similar services with lower carbon dioxide emissions, or directly reducing emissions by capturing and sequestering emissions. Technology to capture carbon dioxide is available today, but capturing and sequestering CO2 will increase the cost of electricity production by an estimated 50%–100% compared with today's generating costs. Moreover, an estimated increase of 15%–30% of the primary energy supply needed to deliver these services or goods would be required. Captured carbon dioxide can be sequestered in deep geological formations, either onshore or offshore. Sedimentary basins are the preferred location for carbon dioxide sequestration, since they are known to contain both the porous and permeable sandstone formations needed to sequester CO2 and low-permeability rocks such as shale that can trap CO2 for geological time periods of millions of years. The estimated capacity for sequestering CO2 is large and expected to be sufficient for at least 100 years of needed demand. However, the actual capacity for safe and environmentally benign sequestration remains uncertain, since CO2 sequestration has been employed for little more than a decade and only on a small scale. Nevertheless, the basic technologies for sequestration and performance prediction are mature, building on nearly a century of oil and gas production, natural-gas storage, CO2-enhanced oil recovery (CO2-EOR), and acid gas disposal. Enhancements of these technologies will arise as geological sequestration itself matures – but they are sufficiently developed to initiate sequestration today. Regulatory and legal issues remain to be resolved, including issues such as permits for sequestration-project siting, well drilling, and completion, operational parameters such as maximum injection pressures, ownership of underground power space, supremacy of mineral or groundwater rights, and liability for long-term stewardship. Resolving these issues and gaining support for this approach from the public are likely to be the greatest challenges for implementing CO2 capture and sequestration on a meaningful scale.
Quantifying the Flow of Exergy and Carbon through the Natural and Human Systems
- Richard E Sassoon, Weston A Hermann, I-Chun Hsiao, Ljuba Milkovic, Aaron J Simon, Sally M Benson
-
- Journal:
- MRS Online Proceedings Library Archive / Volume 1170 / 2009
- Published online by Cambridge University Press:
- 31 January 2011, 1170-R01-03
- Print publication:
- 2009
-
- Article
- Export citation
-
Exergy is the useful portion of energy that allows us to do work and perform energy services. While energy is conserved, exergy is not; some exergy is destroyed whenever energy undergoes a conversion. We gather exergy from distinct, energy-carrying resources that are found in the natural world. These resources are converted into energy carriers that are convenient to use in our factories, vehicles, and buildings for heating, lighting and mechanical services. While there is no shortage of exergy resources, there are considerable environmental, economic, and other constraints associated with the manner and magnitude of their use. This article describes an approach to examining and presenting data on energy use at a global scale. It provides insights into the efficiencies and carbon emissions of many energy pathways, and can provide a basis for an examination of future energy options. In this study, we trace the flow of exergy and carbon through the natural and human systems, revealing the major destructions of exergy, the exergy efficiency of engineered energy processes, and the processes with the highest associated atmospheric carbon emissions. These data have been collected in a relational database available online at http://gcep.stanford.edu/research/exergy/data.html and are presented here in a set of exergy and carbon flow charts.
Sustainability and Energy Conversions
- Sally M. Benson, Franklin M. Orr, Jr.
-
- Journal:
- MRS Bulletin / Volume 33 / Issue 4 / April 2008
- Published online by Cambridge University Press:
- 31 March 2011, pp. 297-302
- Print publication:
- April 2008
-
- Article
-
- You have access Access
- Export citation
-
A sustainable global energy system requires a transition away from energy sources with high greenhouse emissions. Vast energy resources are available to meet our needs, and technology pathways for making this transition exist. Lowering the cost and increasing the reliability and quality of energy from sustainable energy sources will facilitate this transition. Changing the world's energy systems is a huge challenge, but it is one that can be undertaken now with improvements in energy efficiency and with continuing deployment of a variety of technologies. Numerous opportunities exist for research in material sciences to contribute to this global-scale challenge.
Carbon Dioxide Capture and Storage
- Sally M. Benson, Franklin M. Orr, Jr.
-
- Journal:
- MRS Bulletin / Volume 33 / Issue 4 / April 2008
- Published online by Cambridge University Press:
- 31 January 2011, pp. 303-305
- Print publication:
- April 2008
-
- Article
-
- You have access Access
- Export citation
-
Reducing CO2 emissions from the use of fossil fuel is the primary purpose of carbon dioxide capture and storage (CCS). Two basic approaches to CCS are available.1,2 In one approach, CO2 is captured directly from the industrial source, concentrated into a nearly pure form, and then pumped deep underground for long-term storage (see Figure 1). As an alternative to storage in underground geological formations, it has also been suggested that CO2 could be stored in the ocean. This could be done either by dissolving it in the mid-depth ocean (1–3 km) or by forming pools of CO2 on the sea bottom where the ocean is deeper than 3 km and, consequently, CO2 is denser than seawater. The second approach to CCS captures CO2directly from the atmosphere by enhancing natural biological processes that sequester CO2 in plants, soils, and marine sediments. All of these options for CCS have been investigated over the past decade, their potential to mitigate CO2 emissions has been evaluated,1 and several summaries are available.1,3,4